
News OEM
Increasing our knowledge of BSA for immunoassay use
October 5, 2020 - Spain

Biokit Innovation team gets ahead to solve critical challenges that we may face when developing immunoassays for our partners
In Vitro Diagnostics are playing a central role in current healthcare systems. Early diagnosis is key to better patient outcomes, and for this reason current health systems are moving from treatment to prevention. In order to keep the pace in this ecosystem, our partners must provide practitioners and their patients with innovative, reliable and high-quality immunoassays in a sustainable and efficient way.
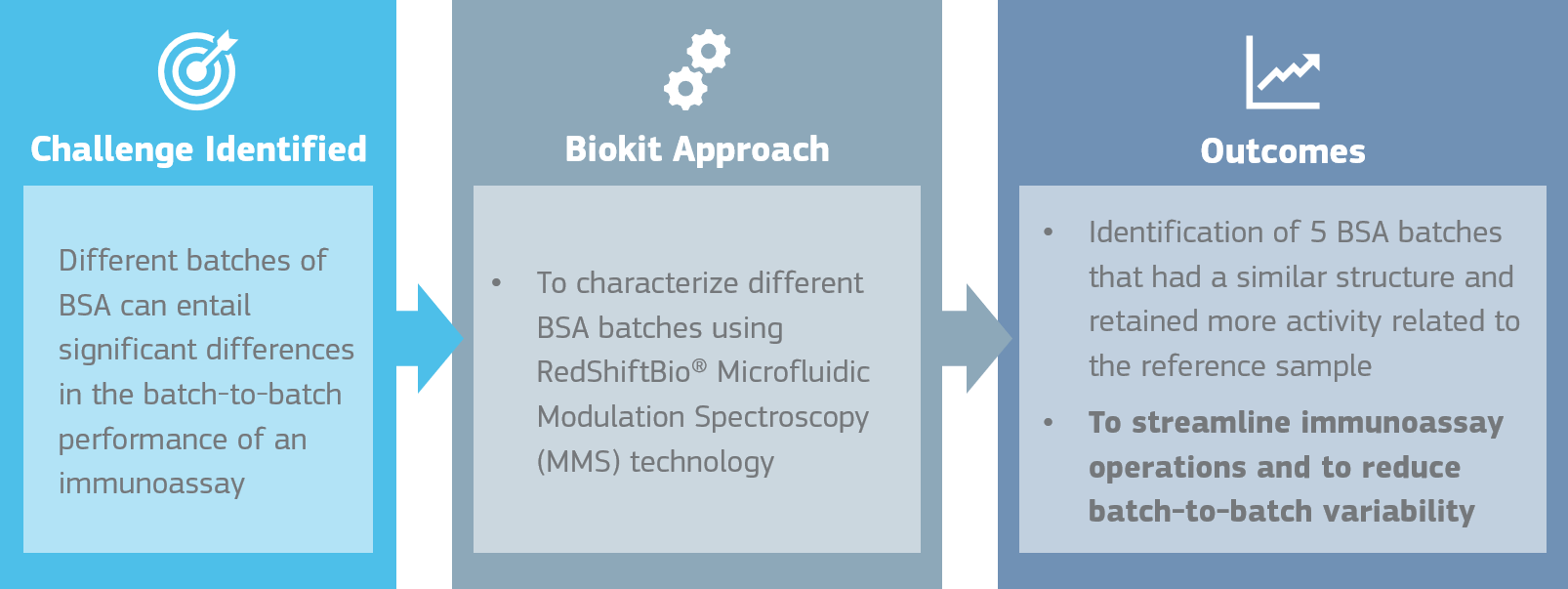
One critical challenge that we must face when developing and manufacturing a new immunoassay is the sourcing of raw materials, and one of the most critical raw materials, apart from the IVD Biomaterial, is the Bovine Serum Albumin when used either as a blocking agent or in the different immunoassay buffers. Different batches of BSA can entail significant differences in the batch-to-batch performance of an immunoassay if the BSA is not previously well characterized and selected.
In Biokit, we work alongside our partners in order to overcome challenges related to the reliability of key raw materials
Our Approach
In order to improve our knowledge of the BSA when used in an immunoassay, we started a collaboration with RedShiftBio®, an innovative company that provides next-generation protein characterization. The Biokit Innovation and Biotechnology team have characterized different BSA batches from different manufacturers, as well as a control batch of BSA, using RedShiftBio® Microfluidic Modulation Spectroscopy (MMS) technology. This technology allows to distinguish higher order protein structure differences lower than 2% in an automatic and more reproducible way than other traditional methods.
Outcomes
A total of 13 BSA samples from different manufacturers were compared to a known BSA reference sample. After the analysis done with MMS, we could identify 5 BSA samples that were more structurally like the reference and retained more activity related to the reference sample. This method would allow us to streamline immunoassay operations for our partners, since we can predict the batches that are viable for reagent manufacturing as well as reduce potential further batch-to-batch variability.
NEWS
Latest News
Please contact us directly via telephone or with the following form.
Tel. +34 93 860 90 00

